Sex-biased DAT Regulation & Psychostimulant Action
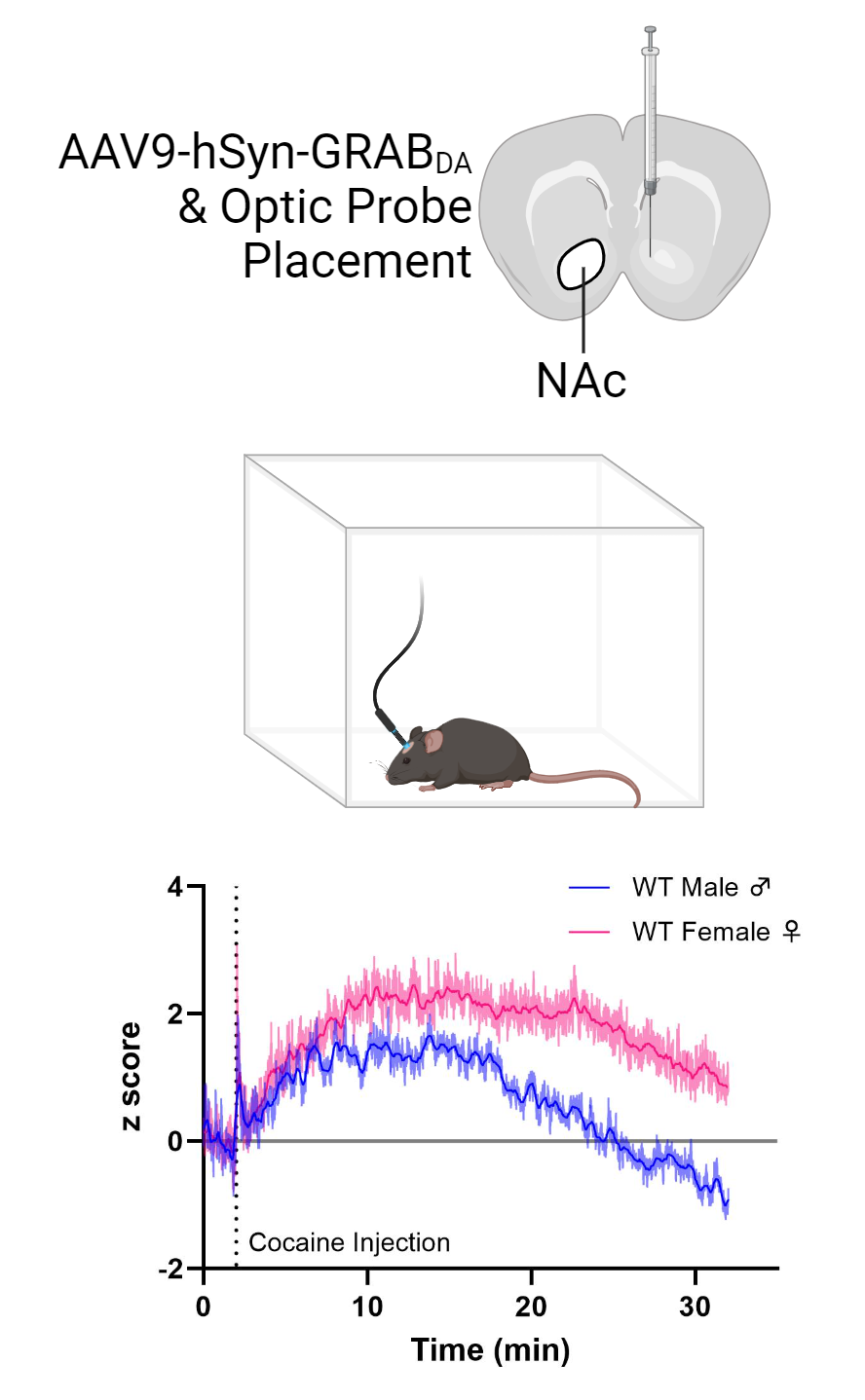
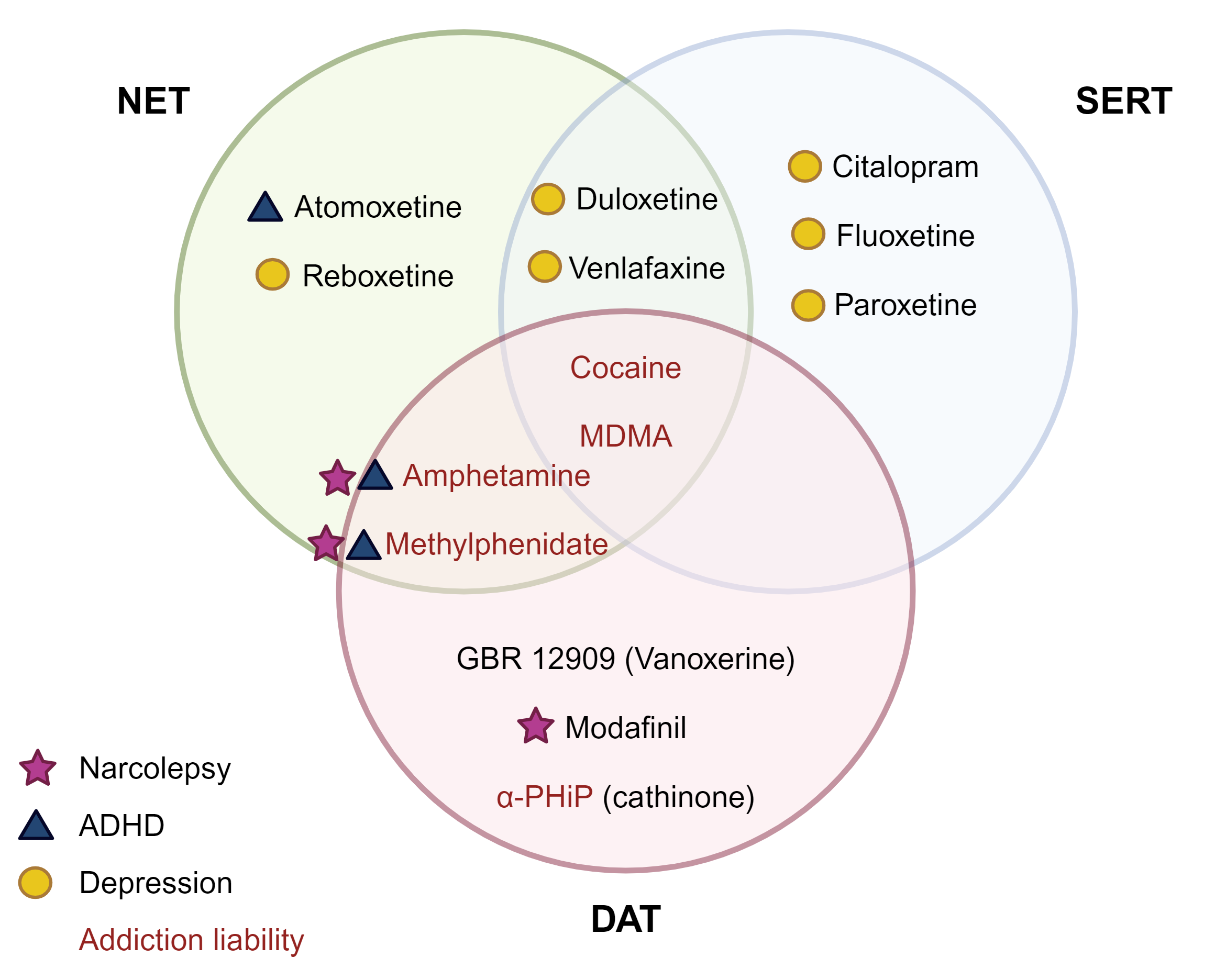
Accumulating evidence demonstrates that genetic/gonadal sex and sex hormones influence several critical brain functions including the release, reuptake, and signaling response to the neurotransmitter dopamine (DA). DA is heavily implicated in substance use disorders and, notably, when compared to men, women typically report experiencing greater subjective effects from psychostimulants, are nearly twice as likely to abuse these drugs, and display enhanced dopamine release in the nucleus accumbens (NAc), a critical component of the mesolimbic dopaminergic reward circuit, following psychostimulant exposure. Similarly, in rodents, there are robust sex differences in the behavioral impacts of psychostimulants such as amphetamine and cocaine. Though existing evidence points to a critical role for gonadal hormones in determining differential psychostimulant responses in females, the neural substrate(s) linking steroid hormone signaling to synaptic DA availability remain unelucidated. We recently described a sex-biased and region-specific functional coupling between the dopamine transporter (DAT), a direct target of psychostimulants and primary mediator of synaptic DA clearance. More specifically, the ability of D2 autoreceptors (D2ARs) to promote phosphorylation and surface trafficking of DAT occurs in the dorsal striatum, the critical efferent target of nigrostriatal dopamine neurons heavily implicated in DA-driven locomotor behavior but is absent in the reward-linked NAc of male mice. In contrast, a functional coupling between D2AR and DAT is observed only in the ventral striatum (NAc) in females.
The goals of this project are threefold:
To establish if/when gonadal hormones and/or sex-specific genetic factors determine sex-biased, region-specific DAT regulation
Determine the neural substrate(s) expressed in a sex-biased and DA neuron-population specific manner that control D2AR-DAT functional coupling
Investigate how circuit-specific DAT regulation shapes DA outflow and psychostimulant-driven behavior in a sex-biased manner
Sex-Specific Consequences of D2R Dysregulation
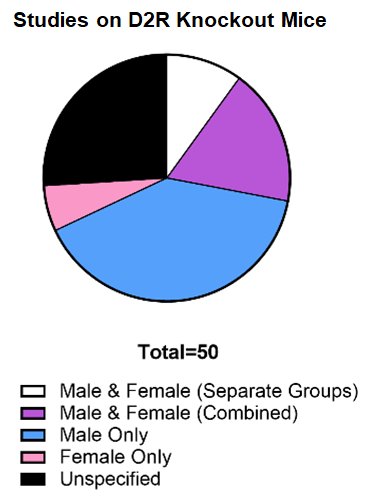
Historically, female subjects have been excluded from preclinical research, with male-only studies in neuroscience research outnumbering those of females by 5.5:1, a discrepancy attributed to concern over the impact of estrus cycling on population homogeneity or the assumption that sexual dimorphism is absent, such that results obtained with males extrapolate to females. Indeed, recent evidence has demonstrated that estrus cycling in female rodents does not contribute to increased variability in metabolic, hormonal, or morphological endpoints relative to males as males also display hormonal heterogeneity due to the formation of dominance hierarchies in group housing. Yet, of the 50 papers published to date characterizing DRD2 knockout or knockdown mice, only 10% analyzed the impact of dopamine D2 receptor (D2R) ablation in both sexes. Deletion of DRD2 from dopaminergic neurons results in hyperactivity, increased motivation, and supersensitivity to cocaine in male mice due to loss of autoreceptor-driven feedback control. Given our data demonstrating that functional coupling between D2ARs and one of its primary effectors (DAT) is both region-specific and sex-biased, it is unlikely that perturbation of this critical signaling pathway will have equivalent consequences in both sexes.
The goals of this project include:
Determining the phenotypic consequence of D2AR deletion on dopamine neurotransmission and behaviors such as threat avoidance, cognition, motivation and sociability linked to dopamine action
Evaluating how perturbation of D2AR function influences development of monoaminergic circuitry
Testing the contribution of D2AR dysregulation to altered behavior in mouse models of psychiatric disorders
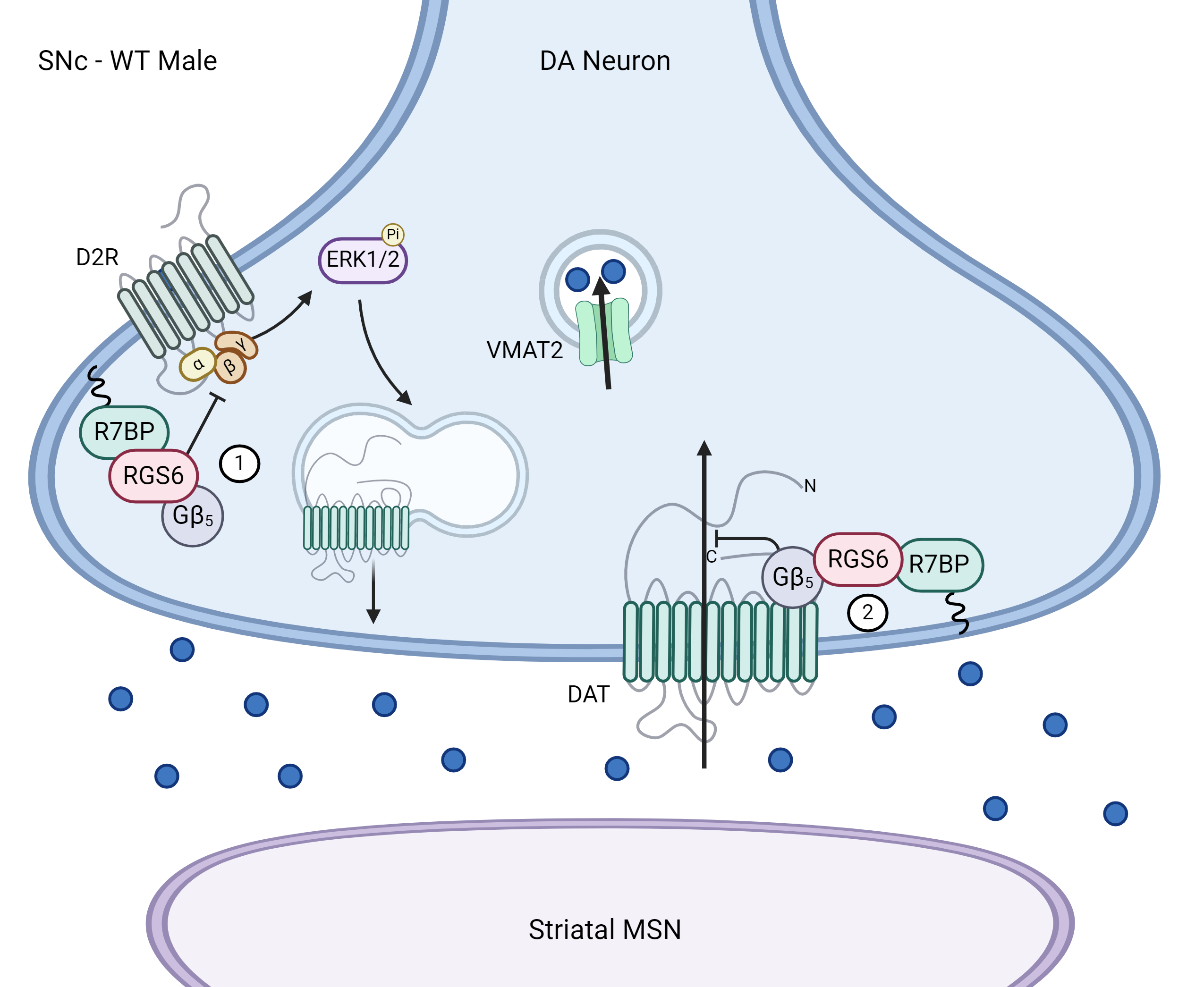
Dopamine Transporter Regulation by Regulator of G protein Signaling 6 (RGS6)
The activity of the dopamine transporter (DAT) is modulated by regulatory mechanisms including a key negative feedback loop mediated by D2-type DA autoreceptors (D2ARs), which promote increased surface trafficking of DAT. Gαi/o-coupled GPCRs such as D2ARs are subject to negative regulation by Regulator of G protein Signaling (RGS) proteins including the R7 family RGS protein RGS6 that stabilizes the transition state in GTP hydrolysis by Gα thereby facilitating termination of G protein signaling. In addition to modulation of D2AR-DAT coupling, RGS6 forms a complex with DAT via recruitment by binding partner Gβ5. Thus, we hypothesize that RGS6 limits dopamine clearance by 1) blocking D2AR-driven DAT surface trafficking and 2) directly impeding dopamine re-uptake. Mice lacking RGS6 display several phenotypes consistent with dopamine dysregulation including reduced alcohol seeking and age-dependent degeneration of dopaminergic neurons that may result from DAT hyperactivity.
The goals of this project include:
Establish the role of RGS6 in DAT regulation
Determine how loss of RGS6-dependent DAT regulation impacts dopaminergic neurotransmission in vivo
Test the involvement of DAT hyperactivity in phenotypes resulting from RGS6 depletion in dopamine neurons